
Neils Bohr Model Limitations of Bohr’s Model of an Atom Bohr discovered that electrons far away from the nucleus had more energy than electrons near nucleus. Rutherford essentially explained how an atom's nucleus works, and Bohr expanded that model to include electrons and their energy levels.Ī tiny (positively charged) nucleus is surrounded by negative electrons traveling in orbits around nucleus in Bohr's concept. Even though early atomic models were erroneous and failed to explain certain experimental results, they served as foundation for future quantum mechanics breakthroughs.Īlso read: Bohr Model of the Hydrogen Atomīohr theory was proposed by Niels Bohr atomic structure model was modified by stating that electrons move in fixed orbitals (shells) and not everywhere in between, and that each orbit (shell) has a fixed energy level.One of the flaws of Rutherford model was that he did not address configuration of electrons in an atom, leaving his theory incomplete.As a result, Rutherford model contradicted Maxwell's theory and was unable to explain atom stability. According to calculations, an electron would disintegrate in nucleus in less than 10-8 seconds using Rutherford model. Electrons would eventually collapse in nucleus.This radiation would transmit energy from electron's motion, but at expense of orbital shrinkage.
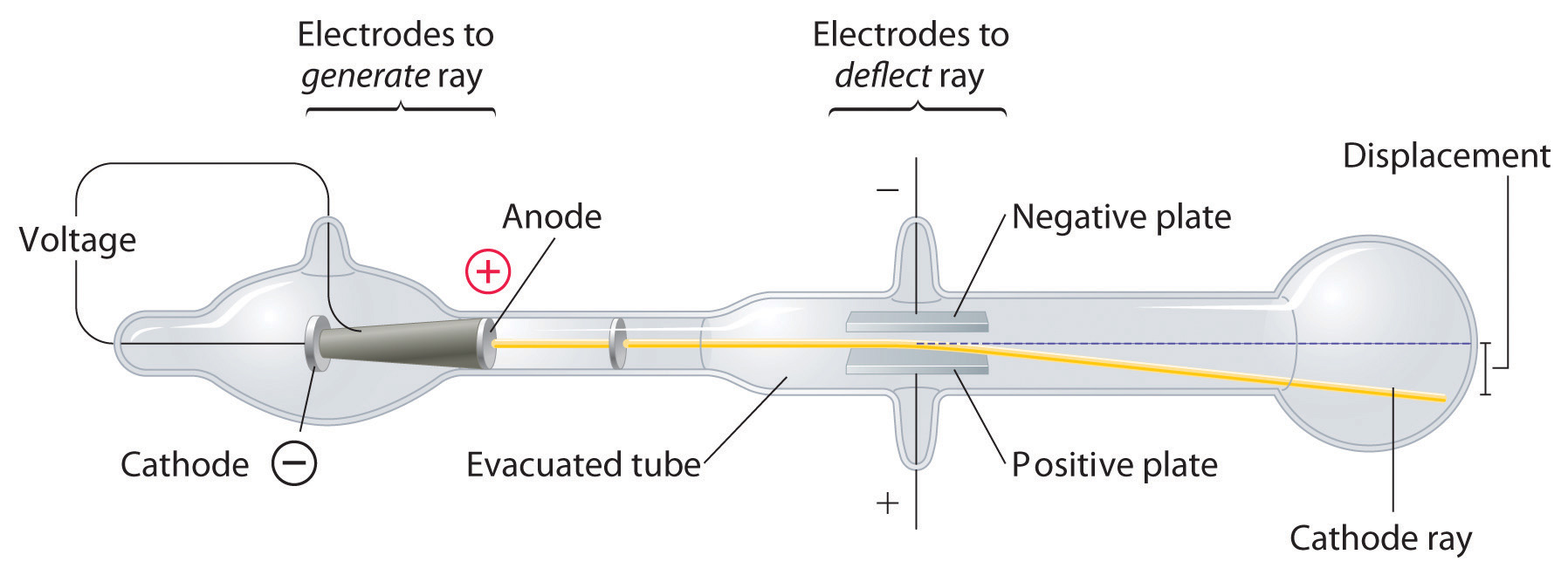
According to Maxwell, accelerating charged particles release electromagnetic radiation, thus an electron rotating around nucleus should do same. According to Rutherford, electrons orbit nucleus in set trajectories termed orbits.Rutherford Experiment Observations Limitations of Rutherford Atomic ModelĮven though Rutherford atomic model was founded on experimental findings, it was unable to explain certain phenomena. As a result, positively charged particles in an atom occupy very little volume in comparison to entire volume of atom. Only a few particles were deflected back, implying that only a few particles had almost 180° deflection angles.In an atom, positive charge is concentrated in a relatively small volume. Gold sheet deflected some particles at minor angles, causing positive charge in each atom to be unevenly distributed.Because a large portion of particles fired at the gold sheet passed through it without being deflected, majority of space in one atom is vacant.Rutherford's observations lead him to following conclusions: Read: Alpha-Particle scattering and Rutherford's Nuclear Model of Atom Observations of Rutherford’s Alpha Scattering Experiment In his experiment, Rutherford focused on high-energy particle streams from a radioactive source at a thin sheet of gold (100 nm thickness). Rutherford carried out an experiment in which he bombarded a thin sheet of gold foil with particles and then analyzed track of these particles after they collided with gold foil. If nucleus of an atom is unbalanced, that is, if number of protons and neutrons differs, atom is unstable. It accomplishes this by emitting particles such as alpha and beta. Process through which unstable nucleus of an atom loses energy by emitting particles is known as radioactivity. However, findings of subsequent trials contradicted its assertions. Atomic model of Thomson was successful in explaining atom's general neutrality. Assumption that atom's mass is equally distributed across atom is key feature of this model. As a result, watermelon model, plum pudding model, and raisin pudding model are all names for this model. The above illustration resembles a chopped watermelon with electrons representing seeds. Electrons are encased in this sphere to create stable electrostatic configuration possible. Thomson proposed that atom is shaped like a sphere with radius of around 10-10m with uniformly distributed positive charge. Cathode rays were found to be negatively charged as a result of this. He discovered that when he used charged metal plates, cathode rays bent away from negative plate and towards positive plate. Thompson used fluorescent coated tube instead of electrometer at one end of Cathode Ray Tube, which would illuminate when cathode ray hit it. He now placed a negatively charged metal plate on one side of cathode rays to allow them to pass through anode and a positively charged metal plate on the other. He then performed a second experiment to determine whether charge carried by cathode rays was negative or positive. He observed that electrometers ceased measuring electric charge after first experiment. Two microscopic perforations in metal led to an electrometer that could measure a modest electric charge. On the other end of his cathode ray tube, he created a metal cylinder. Thompson performed the first cathode ray tube experiment to demonstrate that rays released by an electron cannon are inseparable from latent charge.
